
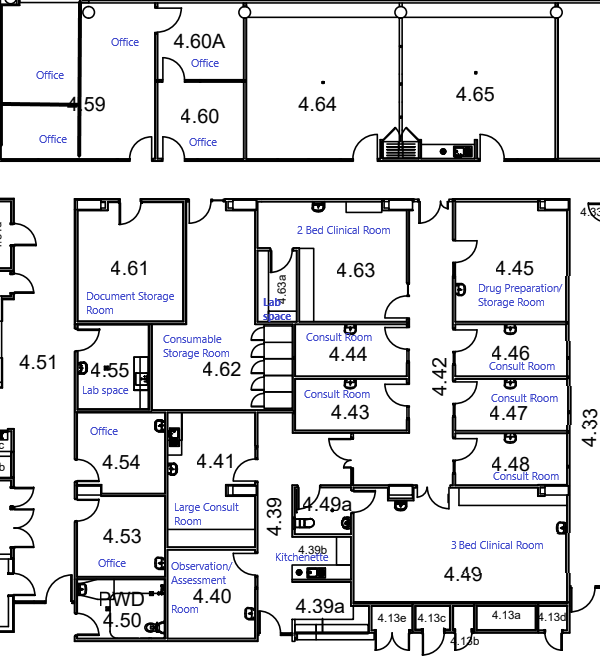
The Clinical Trial Unit (CTU) was established as a Core Research Facility of Griffith University in 2016. It supports Griffith staff and students to conduct their clinical trial research by providing purpose-built, Good Clinical Practice (GCP) aligned clinical trial facilities, as well as offering clinical trial coordination and clinical services to researchers using CTU facilities. The CTU also provides professional clinical trial services to external commercial clients such as the pharmaceutical, biotech, nutraceutical and complementary medicine industries
Services Offered are:
How to access equipment/consultation rooms and services:
1.) Request new project via Request Services tab
2.) Arrange induction and equipment training via Request Services tab
3.) Book rooms and equipment via Schedule Resources tab
4.) Request CTU Clinical Research Nurse time via Schedule Resources tab under Clinical Research Nurses
5.) DRAFT Quote for your project via this link (only accessible for Griffith internal users - please download local copy to edit file)
| Location | Hours |
|
Clinical Trial Unit (G40) Level 4 Gold Coast Campus Southport, Qld, 4214 |
Staffed*: Monday - Friday 8 am - 4 pm |
* Facility is available 24/7 for inducted users.
Click here to see how you can find us on a map
Evelin Tiralongo | Prof |CTU Director | 56780366 | e.tiralongo@griffith.edu.au
Emi Zakar | Business Manager | 56780929 | e.zakar@griffith.edu.au
Elisabeth Kolarich | MSc | Operations Manager | 56780352 | e.kolarich@griffith.edu.au